

Applied Mathematics and Mechanics >
A spinal circuit model with an asymmetric cervical-lumbar layout for limb coordination and gait control in quadrupeds
Received date: 2025-01-19
Revised date: 2025-06-09
Online published: 2025-07-28
Supported by
Project supported by the National Natural Science Foundation of China (Nos. 12272092 and 12332004)
Copyright
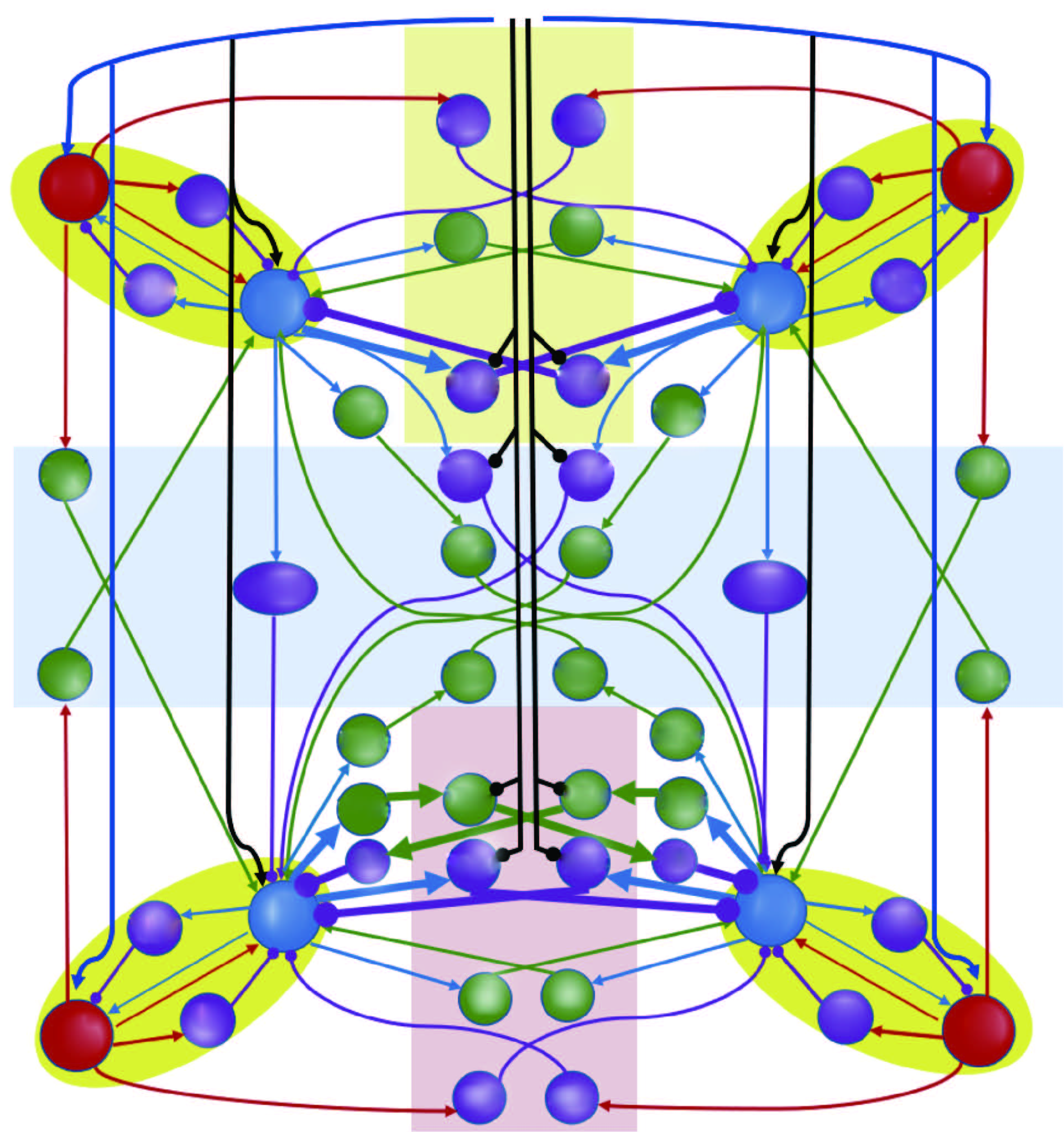
In quadrupeds, the cervical and lumbar circuits work together to achieve the speed-dependent gait expression. While most studies have focused on how local lumbar circuits regulate limb coordination and gaits, relatively few studies are known about cervical circuits and even less about locomotor gaits. We use the previously published models by Danner et al. (DANNER, S. M., SHEVTSOVA, N. A., FRIGON, A., and RYBAK, I. A. Computational modeling of spinal circuits controlling limb coordination and gaits in quadrupeds. eLife, 6, e31050 (2017)) as a basis, and modify it by proposing an asymmetric organization of cervical and lumbar circuits. First, the model reproduces the typical speed-dependent gait expression in mice and more biologically appropriate locomotor parameters, including the gallop gait, locomotor frequencies, and limb coordination of the forelimbs. Then, the model replicates the locomotor features regulated by the M-current. The walk frequency increases with the M-current without affecting the interlimb coordination or gaits. Furthermore, the model reveals the interaction mechanism between the brainstem drive and ionic currents in regulating quadrupedal locomotion. Finally, the model demonstrates the dynamical properties of locomotor gaits. Trot and bound are identified as attractor gaits, walk as a semi-attractor gait, and gallop as a transitional gait, with predictable transitions between these gaits. The model suggests that cervical-lumbar circuits are asymmetrically recruited during quadrupedal locomotion, thereby providing new insights into the neural control of speed-dependent gait expression.
Qinghua ZHU , Fang HAN , Qingyun WANG . A spinal circuit model with an asymmetric cervical-lumbar layout for limb coordination and gait control in quadrupeds[J]. Applied Mathematics and Mechanics, 2025 , 46(8) : 1433 -1450 . DOI: 10.1007/s10483-025-3282-9
| [1] | BATKA, R. J., BROWN, T. J., MCMILLAN, K. P., MEADOWS, R. M., JONES, K. J., and HAULCOMB, M. M. The need for speed in rodent locomotion analyses. Anatomical Record, 297(10), 1839–1864 (2014) |
| [2] | BELLARDITA, C. and KIEHN, O. Phenotypic characterization of speed-associated gait changes in mice reveals modular organization of locomotor networks. Current Biology, 25(11), 1426–1436 (2015) |
| [3] | LEIRAS, R., CREGG, J. M., and KIEHN, O. Brainstem circuits for locomotion. Annual Review of Neuroscience, 45, 63–85 (2022) |
| [4] | BROCARD, F. New channel lineup in spinal circuits governing locomotion. Current Opinion in Physiology, 8, 14–22 (2019) |
| [5] | GRILLNER, S. and EL MANIRA, A. Current principles of motor control, with special reference to vertebrate locomotion. Physiological Reviews, 100, 271–320 (2020) |
| [6] | WILSON, A. C. and SWEENEY, L. B. Spinal cords: symphonies of interneurons across species. Frontiers in Neural Circuits, 17, 1146449 (2023) |
| [7] | CAGGIANO, V., LEIRAS, R., GO?I-ERRO, H., MASINI, D., BELLARDITA, C., BOUVIER, J., CALDEIRA, V., FISONE, G., and KIEHN, O. Midbrain circuits that set locomotor speed and gait selection. nature, 553(7689), 455–460 (2018) |
| [8] | JOSSET, N., ROUSSEL, M., LEMIEUX, M., LAFRANCE-ZOUBGA, D., RASTQAR, A., and BRETZNER, F. Distinct contributions of mesencephalic locomotor region nuclei to locomotor control in the freely behaving mouse. Current Biology, 28(6), 884–901 (2018) |
| [9] | VERNEUIL, J., BROCARD, C., TROUPLIN, V., VILLARD, L., PEYRONNET-ROUX, J., and BROCARD, F. The M-current works in tandem with the persistent sodium current to set the speed of locomotion. PLoS Biology, 18(1), e3000738 (2020) |
| [10] | DROUILLAS, B., BROCARD, C., ZANELLA, S., BOS, R., and BROCARD, F. Persistent Nav1.1 and Nav1.6 currents drive spinal locomotor functions through nonlinear dynamics. Cell Reports, 42, 113085 (2023) |
| [11] | SVENSSON, E., WILLIAMS, M. J., and SCHI?TH, H. B. Neural cotransmission in spinal circuits governing locomotion. Trends in Neurosciences, 41(8), 540–555 (2018) |
| [12] | C?Té, M. P., MURRAY, L. M., and KNIKOU, M. Spinal control of locomotion: individual neurons, their circuits and functions. Frontiers in Physiology, 9, 784 (2018) |
| [13] | HAQUE, F. and GOSGNACH, S. Mapping connectivity amongst interneuronal components of the locomotor CPG. Frontiers in Cellular Neuroscience, 13, 443 (2019) |
| [14] | TALPALAR, A. E., BOUVIER, J., BORGIUS, L., FORTIN, G., PIERANI, A., and KIEHN, O. Dual-mode operation of neuronal networks involved in left-right alternation. nature, 500(7460), 85–88 (2013) |
| [15] | KIEHN, O. Decoding the organization of spinal circuits that control locomotion. Nature Reviews Neuroscience, 17(4), 224–238 (2016) |
| [16] | GRILLNER, S. and KOZLOV, A. The CPGs for limbed locomotion-facts and fiction. International Journal of Molecular Sciences, 22(11), 5882 (2021) |
| [17] | FRIGON, A. The neural control of interlimb coordination during mammalian locomotion. Journal of Neurophysiology, 117(6), 2224–2241 (2017) |
| [18] | RUDER, L., TAKEOKA, A., and ARBER, S. Long-distance descending spinal neurons ensure quadrupedal locomotor stability. Neuron, 92(5), 1063–1078 (2016) |
| [19] | POCRATSKY, A. M., SHEPARD, C. T., MOREHOUSE, J. R., BURKE, D. A., RIEGLER, A. S., HARDIN, J. T., BEARE, J. E., HAINLINE, C., STATES, G. J., BROWN, B. L., WHITTEMORE, S. R., and MAGNUSON, D. S. Long ascending propriospinal neurons provide flexible, context-specific control of interlimb coordination. eLife, 9, e53565 (2020) |
| [20] | ZHANG, J., LANUZA, G. M., BRITZ, O., WANG, Z., SIEMBAB, V. C., ZHANG, Y., VELASQUEZ, T., ALVAREZ, F. J., FRANK, E., and GOULDING, M. V1 and V2b interneurons secure the alternating flexor-extensor motor activity mice require for limbed locomotion. Neuron, 82(1), 138–150 (2014) |
| [21] | CABELGUEN, J. M., ORSAL, D., PERRET, C., and ZATTARA, M. Central pattern generation of forelimb and hindlimb locomotor activities in the cat. Advances in Physiology Education, 1, 199–211 (1980) |
| [22] | HAYASHI, M., HINCKLEY, C. A., DRISCOLL, S. P., MOORE, N. J., LEVINE, A. J., HILDE, K. L., SHARMA, K., and PFAFF, S. L. Graded arrays of spinal and supraspinal V2a interneuron subtypes underlie forelimb and hindlimb motor control. Neuron, 97(4), 869–884 (2018) |
| [23] | CRONE, S. A., ZHONG, G., HARRIS-WARRICK, R., and SHARMA, K. In mice lacking V2a interneurons, gait depends on speed of locomotion. The Journal of Neuroscience, 29(21), 7098–7109 (2009) |
| [24] | AZIM, E., JIANG, J., ALSTERMARK, B., and JESSELL, T. M. Skilled reaching relies on a V2a propriospinal internal copy circuit. nature, 508(7496), 357–363 (2014) |
| [25] | ZELENIN, P. V., VEMULA, M. G., LYALKA, V. F., KIEHN, O., TALPALAR, A. E., and DELIAGINA, T. G. Differential contribution of V0 interneurons to execution of rhythmic and nonrhythmic motor behaviors. The Journal of Neuroscience, 41(15), 3432–3445 (2021) |
| [26] | DANNER, S. M., WILSHIN, S. D., SHEVTSOVA, N. A., and RYBAK, I. A. Central control of interlimb coordination and speed-dependent gait expression in quadrupeds. Journal of Physiology, 594(23), 6947–6967 (2016) |
| [27] | DANNER, S. M., SHEVTSOVA, N. A., FRIGON, A., and RYBAK, I. A. Computational modeling of spinal circuits controlling limb coordination and gaits in quadrupeds. eLife, 6, e31050 (2017) |
| [28] | AUSBORN, J., SHEVTSOVA, N. A., CAGGIANO, V., DANNER, S. M., and RYBAK, I. A. Computational modeling of brainstem circuits controlling locomotor frequency and gait. eLife, 8, e43587 (2019) |
| [29] | LEMIEUX, M., JOSSET, N., ROUSSEL, M., COURAUD, S., and BRETZNER, F. Speed-dependent modulation of the locomotor behavior in adult mice reveals attractor and transitional gaits. Frontiers in Neuroscience, 10, 42 (2016) |
| [30] | ZHU, Q., WANG, F., and HAN, F. Contributions of M- and persistent sodium currents in regulating locomotor rhythms: a computational modeling study. International Journal of Bifurcation and Chaos, 33(12), 2350143 (2023) |
| [31] | ZHU, Q., HAN, F., YU, Y., WANG, F., WANG, Q., and SHAKEEL, A. A spinal circuit model with asymmetric cervical-lumbar layout controls backward locomotion and scratching in quadrupeds. Neural Networks, 178, 106422 (2024) |
| [32] | SHEVTSOVA, N. A., LI, E. Z., SINGH, S., DOUGHERTY, K. J., and RYBAK, I. A Ipsilateral and contralateral interactions in spinal locomotor circuits mediated by V1 neurons: insights from computational modeling. International Journal of Molecular Sciences, 23, 5541 (2022) |
| [33] | ZHONG, G., SHEVTSOVA, N. A., RYBAK, I. A., and HARRIS-WARRICK, R. M. Neuronal activity in the isolated mouse spinal cord during spontaneous deletions in fictive locomotion: insights into locomotor central pattern generator organization. The Journal of Physiology, 590(19), 4735–4759 (2012) |
| [34] | MACHADO, T. A., PNEVMATIKAKIS, E., PANINSKI, L., JESSELL, T. M., and MIRI, A. Primacy of flexor locomotor pattern revealed by ancestral reversion of motor neuron identity. Cell, 162(2), 338–350 (2015) |
| [35] | YANG, L., SINGLA, D., WU, A. K., CROSS, K. A., and MASMANIDIS, S. C. Dopamine lesions alter the striatal encoding of single-limb gait. eLife, 12, RP92821 (2023) |
| [36] | FAN, D., ZHENG, Y., YANG, Z., and WANG, Q. Improving control effects of absence seizures using single-pulse alternately resetting stimulation (SARS) of corticothalamic circuit. Applied Mathematics and Mechanics (English Edition), 41(9), 1287–1302 (2020) https://doi.org/10.1007/s10483-020-2644-8 |
| [37] | ZHU, Q., HAN, F., YUAN, Y., and SHEN, L. A TAN-dopamine interaction mechanism based computational model of basal Ganglia in action selection. Cognitive Neurodynamics, 18, 2127–2144 (2024) |
| [38] | MAO, X. and LEI, F. Stability analysis and coexisting behaviors of a delayed multiplex network under electromagnetic radiation. International Journal of Bifurcation and Chaos, 32(10), 2250148 (2022) |
| [39] | MA, J., QIN, H., SONG, X., and CHU, R. T. Pattern selection in neuronal network driven by electric autapses with diversity in time delays. International Journal of Modern Physics B, 29(1), 1450239 (2015) |
| [40] | JI, Y. and MAO, X. Fast and slow dynamical behaviors of delayed-coupled thermosensitive neurons under electromagnetic induction. Chaos Solitons & Fractals, 189, 115721 (2024) |
| [41] | LEI, Z. and MA, J. Coherence resonance and energy dynamics in a memristive map neuron. Chaos, 35(2), 023158 (2025) |
| [42] | MAO, X., LI, X., DING, W., WANG, S., ZHOU, X., and QIAO, L. Dynamics of a multiplex neural network with delayed couplings. Applied Mathematics and Mechanics (English Edition), 42(3), 441–456 (2021) https://doi.org/10.1007/s10483-021-2709-6 |
/
| 〈 |
|
〉 |